 
Not to miss out, element 102, Nobelium, No, is named in honor of Alfred Nobel, who set aside his vast fortune to establish Nobel Prizes. Mendeleev never received a Nobel Prize for his work, but element 101 was named Mendelevium, Md, after him. For instance they discovered phosphorus when they isolated it from urine. The noble gases (historically also the inert gases, sometimes referred to as aerogens) are the naturally occurring members of group 18 of the periodic table: helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn). Group 3-12: Transition and Inner transition metals group. 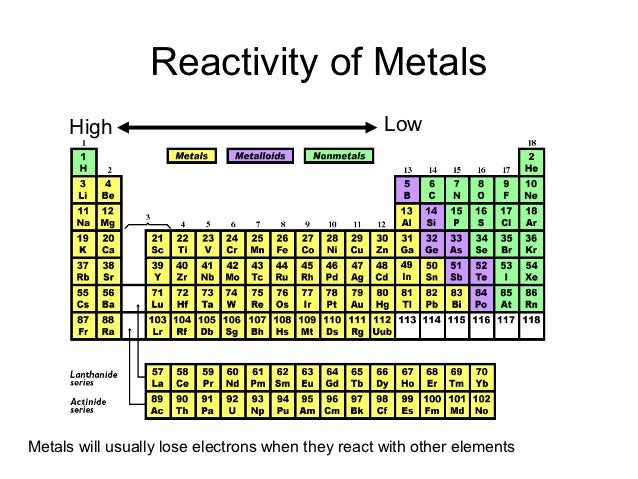
Group 1: Alkali metals group (hydrogen not included) Group 2: Alkaline earth metals group. Over time these gaps have gradually been filled in as scientists unearthed new elements. There are total 18 different groups in Periodic table. Not only did Mendeleev arrange the elements in the correct way, but he also had the foresight to leave gaps for undiscovered elements. At that time, he had only 50 elements to arrange. He wrote the properties of the elements on pieces of card and rearranged them until he realised that, by putting them in order of increasing atomic weight, certain properties of elements regularly occurred. Then in 1869, a Russian scientist called Dmitri Mendeleev produced one of the first practical periodic tables. Several other attempts were made to group elements together over the coming decades. The earliest attempt to classify the elements was in 1789, when Antoine Lavoisier grouped the elements based on their properties into gases, non-metals, metals and earths. The discovery of other elements followed regularly and soon it became necessary to arrange them in some sort of order. Metals are found on the left and in the middle, whereas. However, the first scientific discovery of an element occurred in 1649 when Hennig Brand discovered phosphorous. The periodic table can be used to find out if an element is a metal or a non-metal. It took a bit of imagination from the French engineer, chemist and inventor, Georges Claude, who early in the 20 th Century first applied an electric discharge to a sealed tube of neon gas. And initially its lack of reactivity meant there were no obvious uses for Neon. Any substance whose valence electrons are partially filled are highly reactive in nature to attain stability.Copper has been used by humans for as much as 7000 years and elements such as gold, silver, tin, lead and mercury have been known for many thousands of years. So a new element in name and nature, finally took its place in the periodic table. 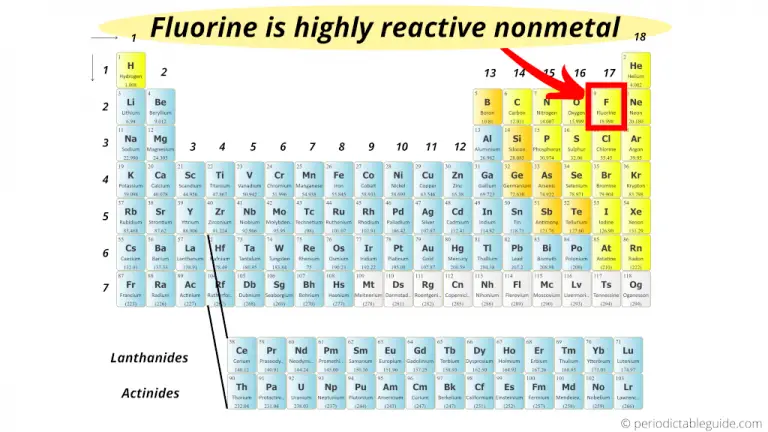
The valence shell of inert gases is filled and hence they can’t be highly reactive in nature. Best Answer Copy All of the elements in Group 18 (0) - Inert Gases (exc. It is an interesting fact that the food is packed in an inert gas to remove oxygen gas and prevents bacteria from growing inside the food. We also know that due to the non-reactive properties of inert gases they are often useful to prevent an undesirable chemical reaction. 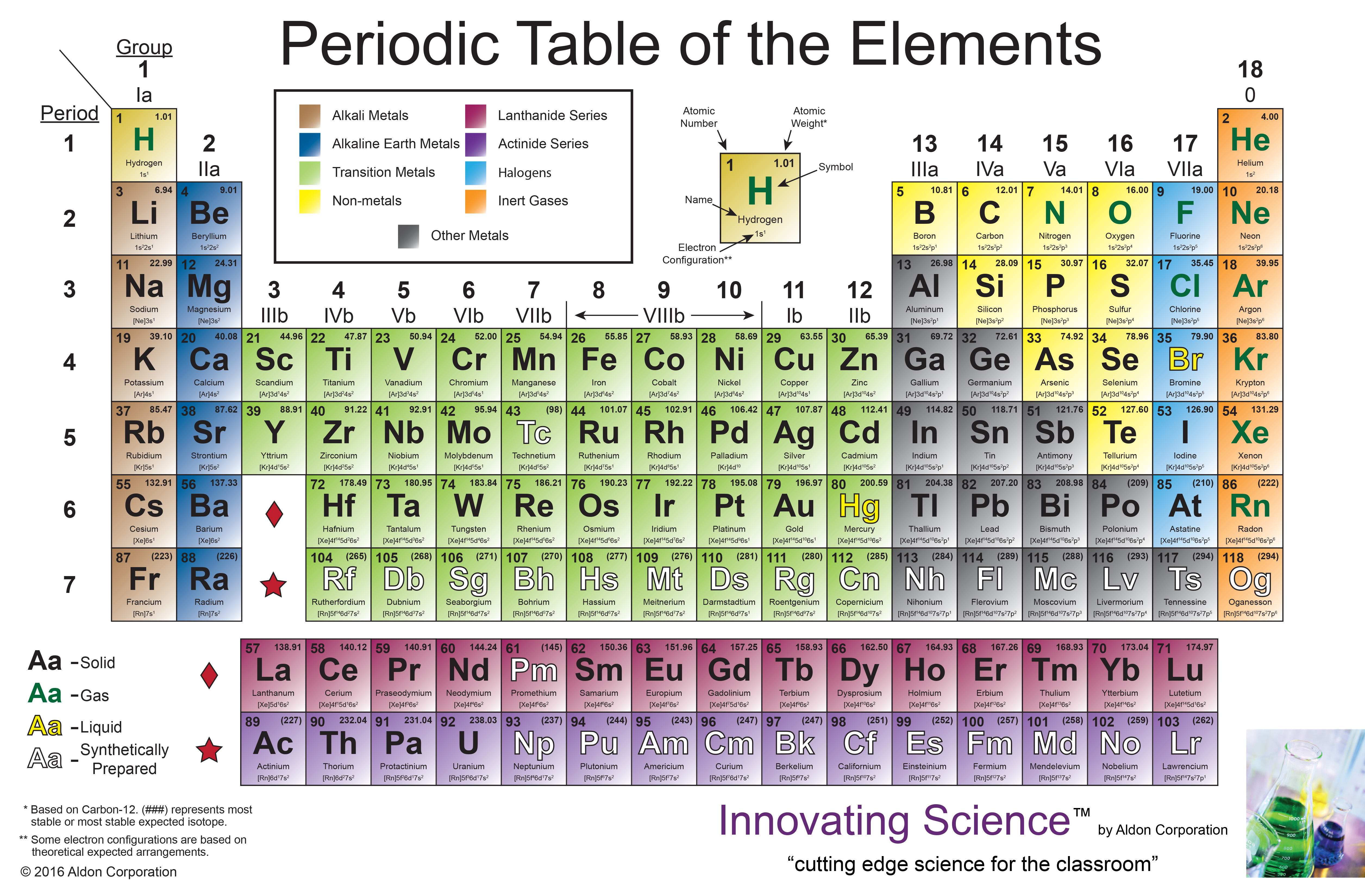
Therefore, we can conclude that the correct answer to this question is Option B.įrom the modern periodic table that the group 18 elements include helium, neon, argon, krypton, xenon, and radon, they are referred to as noble gases or inert gases. Hence, Noble gases are non-reactive in nature. The general electronic configuration of inert of nobles gases are:Īs the noble gas is already stable they do not undergo reaction. So to attain stability they transfer their electrons to form a bond.īut we know in the case of the noble gas the valence shell is completely filled and hence stable in its elemental state. The atoms or molecules react with the transfer of an electron to form a compound.Įlements can donate or accept the electrons because they have partially filled valence electrons and hence unstable. We can also say that inert means not interacting with other molecules. Hint: The gas is called inert which means the atoms or molecules of the particular gas do not combine with the other atoms or molecules. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
